DOI: https://doi.org/10.56669/LWUO5639
ABSTRACT
One of the efforts to support the success of reclamation of ex-silca sand mining land is site quality improvement. The objective of this research was to analyze the effect of application of Arbuscular Mycorrhizal Fungi (AMF) and soil ameliorant on the change of chemical properties of ex-silica sand mining land soil and the growth of Calliandra calothyrsus planting stock. The experiment consisted of two factors (AMF and soil ameliorants) with three replications. The first factor comprises two levels (without AMF and with AMF). The second factor comprising six composition soil ameliorant levels (1. Control :0 g compost and 0 g lime; 2.0 g compost and 5.79 g lime; 3.0 g compost and 10.58 g lime; 4. 35 g compost and 0 g lime; 5.35 g compost and 5.79 g lime; 6.35 g compost and 10.58 g lime). Interaction between AMF, compost, and limeincreased soil pH into neutral category and reduced Al content to a low level which was not measurable. Interaction between AMF, compost, and lime, significantly increased height, diameter, biomass, and mycorrhiza colonization on C. calothyrsus. Combination of AMF, 35 g compost and 5.79 g lime was the best treatment. These treatments increased the height and diameter by 1,106.82 % and 563.22% respectively as compared with those of control plants. While the combination of AMF and 35 g compost (A1B3) increased plant biomass by 297.04 % compared to control plant.
Keywords: Arbuscular Mycorrhizal Fungi, Callindra calothyrsus, post mining land, soil ameliorant
INTRODUCTION
Most mining activities are conducted by open system (open-pit mining), which cause scraping of the upper soil surface, digging (cutting) and filling (Ghose and Majee, 2000). The result of this open-pit mining system is the removal of all soil’s upper layers, that will cause deterioration of the physical, chemical and biological properties of the soil (Arshi, 2015). The mining activity also affects some environmental impacts such as development of acidic soils and waters, erosion of tailings by wind and water, and physical disturbances to the landscape (Benidireet al., 2020). Soil with low pH in ex-mining land could retard plant growth by increasing the solubility of heavy metals and toxic elements in soil (Wahab and Marikar, 2012). Acid soil condition could decompose existing Aluminum in the soil. Aluminum acted as a limiting factor in plant growth by inhibiting root growth and root extension so the plants could not extract nutrients from the soil. This will disturb the interaction between nutrient absorption by plants, such as reducing absorption of other essential nutrients (N, P, K, Ca, Mg, Zn, Fe) by plants (Anetorand Akinrinde, 2007). Toxic elements can cause physiological and biochemical disorder during plant growth (Delhaize and Ryan, 1995).
Many researchers have been using soil ameliorants for improving plant growth and development in acidic soil medium (Ohsowskiet al., 2017; Mrabetet al., 2014; Budi et al., 2020). Lime is a common material for increasing soil pH and have been reported by several researchers (Mrabet et al., 2014; Jayani, Budi and Pamoengkas, 2018) and can improve plant growth and development. Compost and biochar have been reported by Ohsowski et al., (2017) for improving plant growth and development in post-mine sandpits. The use of Arbuscular Mycorrhizal Fungi for improving plant growth in post nickel mined soil medium have been reported by several researchers (Husna et al., 2015; Husna et al., 2019). In the rhizosphere, AMF interact positively with biochar and give synergetic effect to the plant growth (Budi and Christina 2013; Budi and Setyaningsih, 2013). Positive interaction of compost, lime and AMF have been reported by Jayani et al., (2018) and can improve early growth of Falcataria moluccana, Samanea saman, and Cassia siamea seedlings grown in post-mine soil medium.
The objectives of this research were to analyze: (1) the improvement of chemical properties of growth media amended with soil ameliorant, and (2) growth response of C. calothyrsus inoculated by AMF, grown in silica post-mining soil medium amended with soil ameliorant.
MATERIALS AND METHODS
Soil sample collection, soil ameliorant and analysis
The soil media was collected from silica post mining area (0-20 cm depth) at Cibadak, Sukabumi District West Jawa (06o55’18.1” S and 106o47’10.8” E). The soil media was air dried, sieved (2 mm) and autoclaved at 1210C for 1 hour at Silviculture Laboratory. Compost was obtained from the company CV. Laksmi Prima Bogor Indonesia with the trade name of Agro Flower. It had the following characteristics: pH 7.71; C-organic 48.79%; N 0.58%; K 1.00% and P205 2.90% that was analyzed at the Soil Laboratory, Department of Soil Science and Land Resources, Faculty of Agriculture Bogor Agriculture University. Lime was purchased from the farmers’ market as dolomite.
Seedlings and AMF preparation
Seeds of C. calothyrsus were originally from the Center of Research and Development for Forest Seed, Bogor Indonesia. Seeds were soaked in hot water (100°C) for 5 minutes. Afterwards, they were soaked in cool water for 24 hours for breaking their dormancy. The seeds were sown in germinator box filled with sterilized zeolite and placed in green house for two weeks. The Arbuscular Mycorrhizae Fungi (AMF) Glomus mosseae were originally from the collection of Silviculture Laboratory, Faculty of Forestry and Environment, Bogor Agriculture University. The fungi were multiplied on kudzu (Pureria javanica) for eight weeks under greenhouse conditions on sterilized zeolite medium.
AMF inoculation
Two weeks old uniform C. Calothyrsus seedlings were transplanted into 500 ml polybags filled with sterilized mixed silica post mining soil and soil ameliorant. Five grams of fresh AMF inoculum containing approximately 50 spores, mycelium, and mycorrhizal roots were placed near the root’s seedling at transplanting (Budi et al., 2020). Uninoculated C. calothyrsus seedlings were prepared as control. Plants were placed in the green house for 12 weeks and watered as needed.
Parameter measurement and harvesting
Plant height (cm) was measured from plant base to upper tip of the plant using meter rule. Plant steam diameter (mm) was measured at 1 cm above the base of the seedling’s stems using digital caliper. The plant roots were sampled before harvest and stained according to the procedure of Clapp et al., (1996). Percentage of root colonization was determined according to the method of O’connor et al., (2001). Sixteen weeks after planting, plants were harvested to determine their biomass (g). Biomass weights were recorded after drying at 70°C for 72 hours until constant weight is attained. The chemical properties of growing media (pH, C-organic, N total, available P and Aluminium content) after 16 weeks of planting were analyzed.
Experiment design
The factorial design was used in this research with two factors namely AMF and Soil ameliorant. The AMF (A) consist of two levels (A0 = uninoculated, A1 = inoculated with Glomus mossease), and soil ameliorants (B) have 6 levels (B0 = Lime 0 g and Compost 0 g; B1= Compost 0 g and lime 5.79 g (equivalences to 23.16 ton/ha); B3= Compost 0 g and lime 10.58 g (equivalences to 42.32 ton/ha); B4= Compost 35 g (equivalences to 140 ton/ha) and lime 0 g; B5= Compost 35 g and lime 5.79 g; B6= Compost 35 g and lime 10.58 g) per pot. Each pot contain 500 g of growing soil media. The experiment was arranged in a completely randomized design in a polybag culture with 3 replicates. All data were treated with two-way analysis of variance (ANOVA). Means were compared using the Duncan’s test (p<0.05).
RESULTS
Chemical properties of growing media
The chemical properties of growing media from silica post mining land are presented in Table 1. It can be seen that soil chemical properties of growing media (Table 1) could not support plant growth and development due to its pH and low to very low levels of essential nutrients concentration in soil medium. On the other hand, some heavy metals such as aluminum and Fe are very high. The soil growing media amended with soil ameliorant, either alone or in combination could increase their pH, available P and reduce their aluminums content (Table 2).

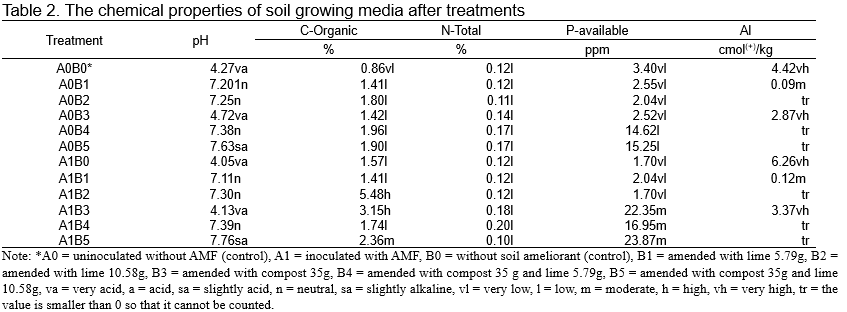
The soil growing media amended with lime 5.79 g (A0B1) and lime 10.58 g (A0B2) increased their pH to the category of slightly acid to neutral and reduced their content of Al to nontoxic level. The addition of lime 5.79 g and compost (A0B4) and lime 10.58 g and compost (A0B5) increased the soil pH to neutral category, and reduce Al content to very low level which could not be measured.
Plant growth
After 16 weeks of planting under greenhouse conditions, the AMF inoculation significantly increased the growth of C. calothyrsus seedling for height, diameter, biomass dry weight and AMF colonization, respectively (Table 3). The application of soil ameliorants and its combination with AMF inoculation significantly increased the growth of C. calothyrsus for height, diameter and biomass after 16 weeks of planting. No significant difference was observed for AMF colonization.

AMF inoculation without combination with soil ameliorant significantly reduced the growth of C. calothyrsus seedling for height, diameter, biomass dry weight respectively after 16 weeks culture in the greenhouse conditions compared to plants grown in soil media amended with soil ameliorants (Table 4).
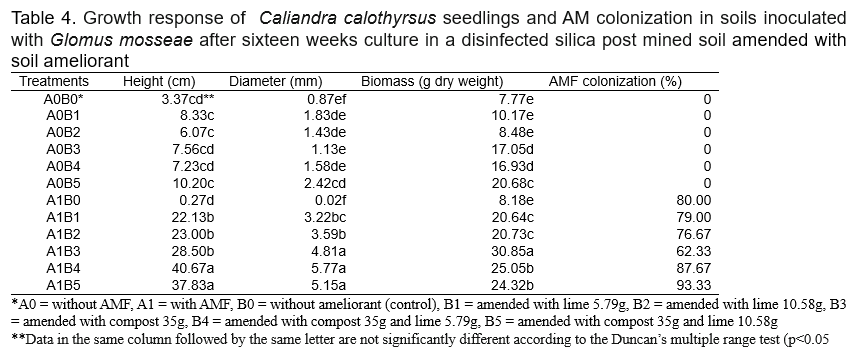
Addition of compost and lime alone or in combination followed by inoculation of plant with AMF significantly increased the growth of C. calothyrsus for height, diameter, biomass dry weight respectively compared to uninoculated control plants. Addition of combination soil ameliorant lime and compost also increased AMF colonization. No AMF structures (internal hypae, arbusculus or vesicle) were observed in uninoculated plants.The highest growth of C. calothyrsuswas shown in plants inoculated with AMF G. mosseae combined with compost 35 g and lime 5.79 g (A1B4). This treatmentincreased height and diameter growth by 1,106.82 % and 563.22%, respectively compared to control plants (Table 4). While the greatest biomass growth was shown in plants inoculated with AMF G. mosseae combined with compost 35 g without lime (A1B3) and increased biomass growth by 297.04% compared to control plants.
DISCUSSION
The results of this study show that: (i) the addition of the soil ameliorant compost or lime alone or in combination to growing soil media originally from silica post mined land, significantly modified the soil pH and soil nutrient contents, (ii) AMF inoculation with G. mosseae significantly improved C. calothyrsus growth after 16 weeks of planting in greenhouse condition and improved chemical soil characteristics when combined with soil ameliorants, and (iii) these changes were significantly linked to a higher mycorrhizal colonization.
Impact of soil ameliorants on soil chemical characteristics
It’s well known that aluminum is a micronutrient needed by plant in small amount and plays an important role as enzyme co-factors in metabolisms processes (Zattaet al., 2000). Aluminum availability in the soil is greatly affected by soil pH (Dong et al., 1995). High aluminum concentration in the soil becomes toxic for plant growth (Delhaize and Ryan, 1995). Numerous studies have shown that several soil ameliorants such as lime and compost can be used for increasing soil pH, reducing soil aluminum content and modified soil nutrients (Anetor and Akinrinde, 2007; Chung, Wu and Ho, 2008; Muindi et al., 2015; Teshome et al., 2017; Novak et al., 2018). Similar results were obtained in our studies that revealed the addition of lime or compost alone or in combination increased soil pH from very acid level to neutral level (Table 2). Interestingly AMF inoculation combined with addition of compost alone (A1B3) or in combination with lime (A1B4 and A1B5) greatly increased availability of soil P (Table 2). AMF have the ability to produce phosphatase enzyme which can alter phosphate compound in the soil into ions and facilitate the plant roots to reabsorb them easily (Bini et al., 2017; Foo et al., 2013). According to Gardner, (2008) soil pH is the main factor that influences solubility and availability of plant nutrients in the soil. Our studies also demonstrated that addition of lime and compost alone or in combination greatly decreased aluminum in soil medium (Table 2).
Impact of soil ameliorants on AMF development and plant growth
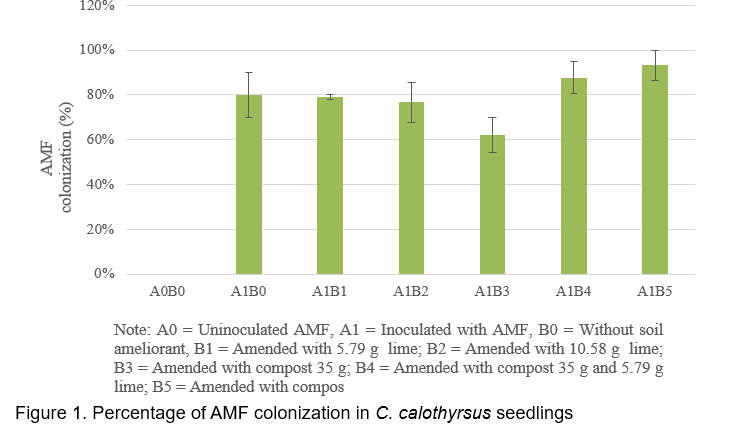
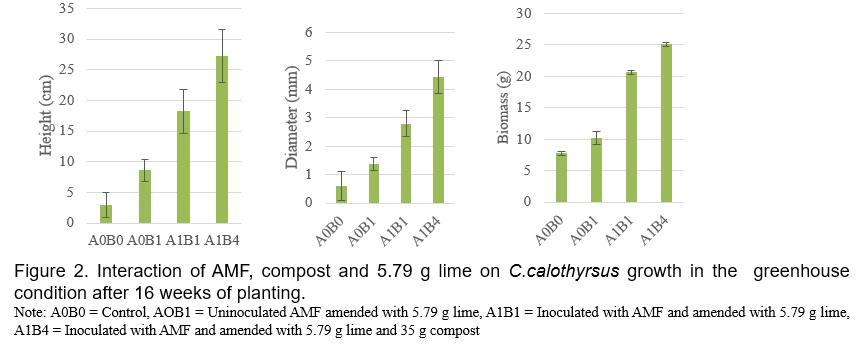
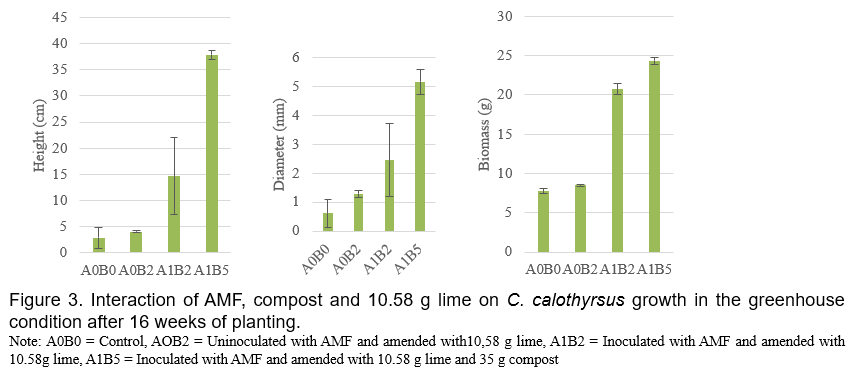
It’s has been demonstrated that AMF is made up of symbiotic organisms between fungi and plant roots which have numerous benefits to plant growth and development. These fungi can increase the uptake of several essential nutrients such as Phosphours (Smith et al., 2011), Nitrogen and Potassium (Bucking and Kafle, 2015; Sharma et al., 2017), producing plant growth hormones and phosphatase enzyme (Bini et al., 2017; Foo et al., 2013), defending root against soil borne diseases (Bakhtiar et al., 2010 ). AMF also interact positively with coal waste and wood charcoal in the mycorrhizosphere to increased plant growth (Budi and Christina, 2013; Budi and Setyaningsih, 2013). In the present study, the addition of compost (A1B3) slightly decreased AMF roots colonization of C. calothyrsus while when compost combined with lime (A1B4 and A1B5) greatly increased AMF roots colonization of C. calothyrsus (Figure 1). The inhibition of AMF development by compost probably due to compost cannot reduce the Aluminum content in soil media as shown in Table 2, the concentrations of Al-dd are very high (3.37 cmol(+)/kg). These results are in accordance with other studies where it has been shown that high Aluminium content inhibit AMF development (Budi et al., 2020). Several studies showed that there are no positive correlations between roots AMF colonization and plant growth. According to Siqueira and Saggin Júnior (1995), soils with low fertility limit plant development and increase the dependence of plants on mycorrhizal association. Under these conditions, fungi grow more extensively inside the root to support the development and functioning of external hyphae (Sanders et al., 1977). In the present study, the growth (height, diameter and biomass) of C. Calothyrsus were lower when inoculated with AMF compared to plant inoculated with AMF combined with soil ameliorants (Figure 2). The soil medium amended with lime 5.79 g without AMF inoculation (A0B1), increased height, diameter and biomass growth of C.calothyrsus by 147.2%; 110.34% and 30.89%, respectively compared to control plant (A0B0). When lime was combined with AMF inoculation (A1B1), growth responses were greater than without AMF inoculation and increased height, diameter and biomass growth of C.calothyrsus by 556.7%, 270.11% and 165.40% respectively compared to control plant (A0B0). The combination of compost 35 g and lime 5.79 g to the soil growth plant medium followed by AMF inoculation (A1B4) significantly increased height, diameter and biomass growth of C.calothyrsus by 1,106.82%; 563.22% and 222.39%, respectively compared to control plants (A0B0), and is the highest among the others combination treatments (Figure 2). From the present study, it is clearly demonstrated that AMF inoculation synergistically increased C. calothyrsus growth if combined with lime alone or in combination with compost. Phosphorus is an essential nutrient for plant growth but in acid soil medium Phosphorus is bound by Aluminum and cannot be absorbed by the roots of the plant. Lime plays an important role in increasing soil pH and reducing Aluminum toxicity in soil medium (Teshome et al., 2017). Furthermore, iron phosphate can be released through an enzymatic phosphatase process which is released by the AMF (Sharma et al., 2017). Similar results were found if the lime dosage is 10.58 g added to soil medium (Figure 3).The soil medium amended with lime 10.58 g without AMF inoculation (A0B2), increased height, diameter and biomass growth of C. Calothyrsus by 80.12%; 64.37% and 9.14%, respectively compared to control plants (A0B0). If lime were combined with AMF inoculation (A1B2), growth response was greater than without AMF inoculation, and it increased height, diameter and biomass growth of C. Calothyrsus by 582.49%, 312.64% and 166.80%, respectively compared to control plant (A0B0). The combination of compost 35 g and lime 10.58 g to the soil growth medium followed by AMF inoculation (A1B5) significantly increased height, diameter and biomass growth of C. calothyrsusby 1,022.55%, 491.95% and 213.00%, respectively compared to control plants (A0B0), and was the highest among the other combination treatments. As shown in Table 2, the addition of lime 10.58 g to soil medium increased soil pH from 4.27 to 7.25 and Aluminium content was reduced to undetected amount. Soil medium amanded with lime 10.58 g and compost 35 g increased their pH from 4.27 to 7.76 (Table 2), indicating compost also can increase soil pH and reduce aluminum content to undetected amount. From the present study, it is demonstrated that soil medium amended by lime 5.79 g is better than amended by lime 10.58 g for increasing C. calothyrsus growth without AMF inoculation. The addition of lime 5.79 g and compost 35 g to soil medium followed by AMF inoculation increased the growth of C.calothyrsus better than soil amended by 10.58 g and compost 35 g followed by AMF inoculation.
CONCLUSION
Post mining soil with low pH and high Aluminium content could be improved by soil ameliorants lime and compost with appropriate dosage. The soil ameliorant lime increased soil pH to neutral level and increased C. calothyrsus growth. Addition of lime and compost to soil media followed by AMF inoculation increased significantly the growth of C. calothyrsus. These findings imply the prospective and potential use of AMF as biofertilzer and soil ameliorant for the succesful reclamation of post mining land.
ACKNOWLEDGEMENT
The authors thank Directorate for Research and Community Services, Ministry of Research, Technology, and Higher Education, the Republic of Indonesia, for providing the funding of this research. The authors also thank PT Holcim Indonesia Tbk., Sukabumi, West Java, for providing soil materials in this research.
REFERENCES
Anetor,M.O., and E.A. Akinrinde, 2007. Lime effectiveness of some fertilizers in a tropical acid alfisol. Journal of Central Europeean Agriculture, 8(1): 17-24.
Arshi A. 2015. Reclamation of coal mine overburden dump through enviromental friendly method. Saudi Journal of Biological Sciences: 24(2017)371-378 doi: 10.1016/j.sjbs.2015.09.009
Bakhtiar, Y., S. Yahya, W.Sumaryono, M.S. Sinaga, S.W. Budi, andT. Tajudin. 2010. Isolation and identification of mycorrhizosphere bacteria and their antagonistic effects towards Ganoderma boniense in vitro. Journal of Microbiology Indonesia, 4(2): 96-102.
Benidire L, L,Benidire, A. Boularbah. 2020. Impacts of mining activities on soil properties: case studies from Morocco mine sites. SOIL SCIENCE ANNUAL. 71(4), 395–407. https://doi.org/10.37501/soilsa/133011
Bini, D., C. Alcara dos Santos, M.C.P da Silva, J.A. Bonfirm, andE.J.B.N. Cardoso. 2017. Intercropping Acacia mangium stimulates AMF colonization and soil phosphatase activity in Eucalyptus grandis. Scientia Agricola, 2(2): 102-110.
Bucking, H. andA. Kafle. 2015. Role of arbuscular fungi in the nitrogen uptake of plants: current knowledge and research gaps. Agronomy, 5: 587-612.
Budi S.W, and F.Christina. 2013 Coal waste powder amendment and arbuscular mycorrhizal fungi enhance the growth of jabon (AnthocephaluscadambaMiq) seedling in ultisol soil medium.Journal Tropica Soils18, 59-66
Budi S.W. and L.Setyaningsih. 2013. Arbuscular mycorrhizal fungi and biochar improved early growth of neem (Melia azedarach Linn.) seedling under greenhouse conditions JurnalManajenenHutanTropika19, 103-110
Budi, S.W. C.Wibowo,A. Sukendro and H. S. Bekti. 2020. Growth improvement of Falcataria moluccana inoculated with MycoSilvi grown in post-mining silica sand soil amended with soil ameliorants. Biodiversitas, 21(1): 422-427
Clapp J.P, A.H Fitter,and J.W.Merryweather. 1996. Arbuskular mycorrhizas. In: Hall GS, Lasserre P, Hawksworth DL, Editor. Methods for the Examination of Organismal Diversity in Soils and Sediments. Wallingford, Oxon (UK): CAB International.
Chung, Ren-shih, Wu, and Sun-ho. 2008. Effect of corncob compost on plant growth in an acid red soil. Journal Communications in Soil science and Plant Analysis,28(9): 673-683.
Delhaize, E. andP.R. Ryan. 1995. Aluminum toxicity and tolerance in plants. Plant. Phytol. 107:315-321.
Dong, D. M.H Ramsey, and I. Thornton. 1995. Effect of soil pH on Al availability in soils and its uptake by the soybean plant (Glycine max). Journal of Geochemical exploration. 55(1): 223-230
Estefan G, R. Sommer and J. Ryan. 2013. Methods of soil, plant, and water analysis: A manual for the west Asia and north, 3rd ed. ICARDA, Beirut, Lebanon.
Foo, E., J.J. Ross, W.T. Jones, andJ.B. Reid. 2013. Plant hormones in arbuscular mycorrhizal symbioses: an emergingrole for gibberellins. Annals of Botany,111: 769–779.
Gardner F.P.R.B Pearce, and R.L. Mitchell. 2008. Physiology of Crop Plants. Ames (UK): The Iowa University Press.
Ghose, M.K. and S.R.Majee. 2000. Assessment of dust generation due to opencast coal mining – an Indian case study. Environ. Monit. Assess. 61, 255–263.
Husna H.S.W Budi, I.Mansur and C. Kusmana. 2015. Growth response of kayu kuku (Pericopsismooniana (Thw.) Thw) seedling to indigenous arbuscular mycorrhizal fungi inoculation JurnalPemuliaanHutanTanaman9, 131-148
Husna H., I.Mansur,S.W.Budi, F.D.Tuheteru, A.Arif, E.J.Tuheteru, Albasari. 2019. Effects of arbuscular mycorrhizalfungi and organic material on growth and nutrient uptake by Pericopsismooniana in coal mine Asian.Journal of Plant Sciences18, 101-109
Jayani F.M, S.W Budi, andP. Pamoengkas 2018. Response of forest tree species inoculated with AMF and soil ameliorant addition grown in silica sand. Asian J Agri & Biol. 6(4):556-565
Mrabet S.E, L.Ouahmane,A.E.Mousadik, F.Msanda, Y. Abbas. 2014. The effectiveness of arbuscular mycorrhizal inoculation and bio-compost addition for enhancing reforestation with Argania spinosa in Morocco Open Journal of Forestry4, 14-23
Muindi, E.M, J. Mrema, E. Semu, P.Mtakwa, and C. Gachene. 2015. Effects of lime-aluminium-phosphate interactions on maize growth and yields in acid soils of the Kenya highlands. American Journal of Agriculture and Forestry,3(6): 244-252.
Novak, J.M.,J.A. Ippolito, T.F. Ducey, D.W. Watts, K.A. Spokas, K.M. Trippe, G.C. Sigua, andM.G. Johnson. 2018. Remediation of an acidic mine spoil: Miscanthus biochar and lime amendment affects metal availability, plant growth, and soil enzyme activity. Chemosphere,205: 709-718.
O’Connor P.J, S.E.Smith, F.A. Smith. 2001. Arbuscular mycorrhizal associations in the southern Southern Simpson desert. Aust J Bot. 49:493-499 doi:10.1071/BT00014
Ohsowski B.M., K.Dunfield, J.N. Klironomos,M.M. Hart. 2017. Plant response to biochar, compost, and mycorrhizal fungal amendments in post-mine sandpits Restoration Ecology 26, 63-72
Sanders, F.E.,P.B. Tinker, R.L.B. Black, and S.M. Palmerley. 1977. The development of endomycorrhizal root systems. I. Spread of infection and growth-promoting effects with four species of vesicular-arbuscular endophyte. New Phytologist 78: 553-559
Sharma, N., K. Yadav, and A.Aggarwal. 2017. Role of potassium and arbuscular mycorrhizal fungi in alleviation of water stress on Vigna mungo. Environmental and Experimental Biology, 15: 15-24.
Siqueira, J.O. andO.J. Saggin-Júnior. 1995. The importance of mycorrhizal association in natural low-fertility soils. Pp.240-280. In: Anais do Simpósio Internacional sobre Estresse Ambiental. Belo Horizonte 1992: EMBRAPA/ CNPMS, Sete Lagoas.
Smith, S.E., I. Jakobsen, M.Gronlund, andF.A. Smith. 2011. Roles of Arbuscular Mycorrhizas in Plant PhosphorusNutrition: Interactions between Pathways of PhosphorusUptake in Arbuscular Mycorrhizal Roots Have ImportantImplications for Understanding and Manipulating PlantPhosphorus Acquisition. Plant Physiology, 156: 1050-1057.
Teshome, B., T. Tana, N. Dechassa, and T.N. Singh. 2017. Effect of compost, lime and P on selected properties of acidic soils of assosa. Biology Agriculture and Healthcare. 7(5): 34-44.
Wahab, S.A.A. and F.A. Marikar. 2012. The environmental impact of gold mines: pollution by heavy metals. Central European Journal of Engineering,2: 304-313.
Zatta,P., E. Lain, and C. Cagnolini. 2000. Effect of aluminium on activity of Krebs cycle enzymes and glutamate dehydrogenase in rat brain homogenate. Eur.J.Biochem, 267:3049-3055.
The Growth of Calliandra calothyrsus Inoculated with Arbuscular Mycorrhizae Fungi on Silica Post Mining Media Amended with Soil Ameliorant
DOI: https://doi.org/10.56669/LWUO5639
ABSTRACT
One of the efforts to support the success of reclamation of ex-silca sand mining land is site quality improvement. The objective of this research was to analyze the effect of application of Arbuscular Mycorrhizal Fungi (AMF) and soil ameliorant on the change of chemical properties of ex-silica sand mining land soil and the growth of Calliandra calothyrsus planting stock. The experiment consisted of two factors (AMF and soil ameliorants) with three replications. The first factor comprises two levels (without AMF and with AMF). The second factor comprising six composition soil ameliorant levels (1. Control :0 g compost and 0 g lime; 2.0 g compost and 5.79 g lime; 3.0 g compost and 10.58 g lime; 4. 35 g compost and 0 g lime; 5.35 g compost and 5.79 g lime; 6.35 g compost and 10.58 g lime). Interaction between AMF, compost, and limeincreased soil pH into neutral category and reduced Al content to a low level which was not measurable. Interaction between AMF, compost, and lime, significantly increased height, diameter, biomass, and mycorrhiza colonization on C. calothyrsus. Combination of AMF, 35 g compost and 5.79 g lime was the best treatment. These treatments increased the height and diameter by 1,106.82 % and 563.22% respectively as compared with those of control plants. While the combination of AMF and 35 g compost (A1B3) increased plant biomass by 297.04 % compared to control plant.
Keywords: Arbuscular Mycorrhizal Fungi, Callindra calothyrsus, post mining land, soil ameliorant
INTRODUCTION
Most mining activities are conducted by open system (open-pit mining), which cause scraping of the upper soil surface, digging (cutting) and filling (Ghose and Majee, 2000). The result of this open-pit mining system is the removal of all soil’s upper layers, that will cause deterioration of the physical, chemical and biological properties of the soil (Arshi, 2015). The mining activity also affects some environmental impacts such as development of acidic soils and waters, erosion of tailings by wind and water, and physical disturbances to the landscape (Benidireet al., 2020). Soil with low pH in ex-mining land could retard plant growth by increasing the solubility of heavy metals and toxic elements in soil (Wahab and Marikar, 2012). Acid soil condition could decompose existing Aluminum in the soil. Aluminum acted as a limiting factor in plant growth by inhibiting root growth and root extension so the plants could not extract nutrients from the soil. This will disturb the interaction between nutrient absorption by plants, such as reducing absorption of other essential nutrients (N, P, K, Ca, Mg, Zn, Fe) by plants (Anetorand Akinrinde, 2007). Toxic elements can cause physiological and biochemical disorder during plant growth (Delhaize and Ryan, 1995).
Many researchers have been using soil ameliorants for improving plant growth and development in acidic soil medium (Ohsowskiet al., 2017; Mrabetet al., 2014; Budi et al., 2020). Lime is a common material for increasing soil pH and have been reported by several researchers (Mrabet et al., 2014; Jayani, Budi and Pamoengkas, 2018) and can improve plant growth and development. Compost and biochar have been reported by Ohsowski et al., (2017) for improving plant growth and development in post-mine sandpits. The use of Arbuscular Mycorrhizal Fungi for improving plant growth in post nickel mined soil medium have been reported by several researchers (Husna et al., 2015; Husna et al., 2019). In the rhizosphere, AMF interact positively with biochar and give synergetic effect to the plant growth (Budi and Christina 2013; Budi and Setyaningsih, 2013). Positive interaction of compost, lime and AMF have been reported by Jayani et al., (2018) and can improve early growth of Falcataria moluccana, Samanea saman, and Cassia siamea seedlings grown in post-mine soil medium.
The objectives of this research were to analyze: (1) the improvement of chemical properties of growth media amended with soil ameliorant, and (2) growth response of C. calothyrsus inoculated by AMF, grown in silica post-mining soil medium amended with soil ameliorant.
MATERIALS AND METHODS
Soil sample collection, soil ameliorant and analysis
The soil media was collected from silica post mining area (0-20 cm depth) at Cibadak, Sukabumi District West Jawa (06o55’18.1” S and 106o47’10.8” E). The soil media was air dried, sieved (2 mm) and autoclaved at 1210C for 1 hour at Silviculture Laboratory. Compost was obtained from the company CV. Laksmi Prima Bogor Indonesia with the trade name of Agro Flower. It had the following characteristics: pH 7.71; C-organic 48.79%; N 0.58%; K 1.00% and P205 2.90% that was analyzed at the Soil Laboratory, Department of Soil Science and Land Resources, Faculty of Agriculture Bogor Agriculture University. Lime was purchased from the farmers’ market as dolomite.
Seedlings and AMF preparation
Seeds of C. calothyrsus were originally from the Center of Research and Development for Forest Seed, Bogor Indonesia. Seeds were soaked in hot water (100°C) for 5 minutes. Afterwards, they were soaked in cool water for 24 hours for breaking their dormancy. The seeds were sown in germinator box filled with sterilized zeolite and placed in green house for two weeks. The Arbuscular Mycorrhizae Fungi (AMF) Glomus mosseae were originally from the collection of Silviculture Laboratory, Faculty of Forestry and Environment, Bogor Agriculture University. The fungi were multiplied on kudzu (Pureria javanica) for eight weeks under greenhouse conditions on sterilized zeolite medium.
AMF inoculation
Two weeks old uniform C. Calothyrsus seedlings were transplanted into 500 ml polybags filled with sterilized mixed silica post mining soil and soil ameliorant. Five grams of fresh AMF inoculum containing approximately 50 spores, mycelium, and mycorrhizal roots were placed near the root’s seedling at transplanting (Budi et al., 2020). Uninoculated C. calothyrsus seedlings were prepared as control. Plants were placed in the green house for 12 weeks and watered as needed.
Parameter measurement and harvesting
Plant height (cm) was measured from plant base to upper tip of the plant using meter rule. Plant steam diameter (mm) was measured at 1 cm above the base of the seedling’s stems using digital caliper. The plant roots were sampled before harvest and stained according to the procedure of Clapp et al., (1996). Percentage of root colonization was determined according to the method of O’connor et al., (2001). Sixteen weeks after planting, plants were harvested to determine their biomass (g). Biomass weights were recorded after drying at 70°C for 72 hours until constant weight is attained. The chemical properties of growing media (pH, C-organic, N total, available P and Aluminium content) after 16 weeks of planting were analyzed.
Experiment design
The factorial design was used in this research with two factors namely AMF and Soil ameliorant. The AMF (A) consist of two levels (A0 = uninoculated, A1 = inoculated with Glomus mossease), and soil ameliorants (B) have 6 levels (B0 = Lime 0 g and Compost 0 g; B1= Compost 0 g and lime 5.79 g (equivalences to 23.16 ton/ha); B3= Compost 0 g and lime 10.58 g (equivalences to 42.32 ton/ha); B4= Compost 35 g (equivalences to 140 ton/ha) and lime 0 g; B5= Compost 35 g and lime 5.79 g; B6= Compost 35 g and lime 10.58 g) per pot. Each pot contain 500 g of growing soil media. The experiment was arranged in a completely randomized design in a polybag culture with 3 replicates. All data were treated with two-way analysis of variance (ANOVA). Means were compared using the Duncan’s test (p<0.05).
RESULTS
Chemical properties of growing media
The chemical properties of growing media from silica post mining land are presented in Table 1. It can be seen that soil chemical properties of growing media (Table 1) could not support plant growth and development due to its pH and low to very low levels of essential nutrients concentration in soil medium. On the other hand, some heavy metals such as aluminum and Fe are very high. The soil growing media amended with soil ameliorant, either alone or in combination could increase their pH, available P and reduce their aluminums content (Table 2).
The soil growing media amended with lime 5.79 g (A0B1) and lime 10.58 g (A0B2) increased their pH to the category of slightly acid to neutral and reduced their content of Al to nontoxic level. The addition of lime 5.79 g and compost (A0B4) and lime 10.58 g and compost (A0B5) increased the soil pH to neutral category, and reduce Al content to very low level which could not be measured.
Plant growth
After 16 weeks of planting under greenhouse conditions, the AMF inoculation significantly increased the growth of C. calothyrsus seedling for height, diameter, biomass dry weight and AMF colonization, respectively (Table 3). The application of soil ameliorants and its combination with AMF inoculation significantly increased the growth of C. calothyrsus for height, diameter and biomass after 16 weeks of planting. No significant difference was observed for AMF colonization.
AMF inoculation without combination with soil ameliorant significantly reduced the growth of C. calothyrsus seedling for height, diameter, biomass dry weight respectively after 16 weeks culture in the greenhouse conditions compared to plants grown in soil media amended with soil ameliorants (Table 4).
Addition of compost and lime alone or in combination followed by inoculation of plant with AMF significantly increased the growth of C. calothyrsus for height, diameter, biomass dry weight respectively compared to uninoculated control plants. Addition of combination soil ameliorant lime and compost also increased AMF colonization. No AMF structures (internal hypae, arbusculus or vesicle) were observed in uninoculated plants.The highest growth of C. calothyrsuswas shown in plants inoculated with AMF G. mosseae combined with compost 35 g and lime 5.79 g (A1B4). This treatmentincreased height and diameter growth by 1,106.82 % and 563.22%, respectively compared to control plants (Table 4). While the greatest biomass growth was shown in plants inoculated with AMF G. mosseae combined with compost 35 g without lime (A1B3) and increased biomass growth by 297.04% compared to control plants.
DISCUSSION
The results of this study show that: (i) the addition of the soil ameliorant compost or lime alone or in combination to growing soil media originally from silica post mined land, significantly modified the soil pH and soil nutrient contents, (ii) AMF inoculation with G. mosseae significantly improved C. calothyrsus growth after 16 weeks of planting in greenhouse condition and improved chemical soil characteristics when combined with soil ameliorants, and (iii) these changes were significantly linked to a higher mycorrhizal colonization.
Impact of soil ameliorants on soil chemical characteristics
It’s well known that aluminum is a micronutrient needed by plant in small amount and plays an important role as enzyme co-factors in metabolisms processes (Zattaet al., 2000). Aluminum availability in the soil is greatly affected by soil pH (Dong et al., 1995). High aluminum concentration in the soil becomes toxic for plant growth (Delhaize and Ryan, 1995). Numerous studies have shown that several soil ameliorants such as lime and compost can be used for increasing soil pH, reducing soil aluminum content and modified soil nutrients (Anetor and Akinrinde, 2007; Chung, Wu and Ho, 2008; Muindi et al., 2015; Teshome et al., 2017; Novak et al., 2018). Similar results were obtained in our studies that revealed the addition of lime or compost alone or in combination increased soil pH from very acid level to neutral level (Table 2). Interestingly AMF inoculation combined with addition of compost alone (A1B3) or in combination with lime (A1B4 and A1B5) greatly increased availability of soil P (Table 2). AMF have the ability to produce phosphatase enzyme which can alter phosphate compound in the soil into ions and facilitate the plant roots to reabsorb them easily (Bini et al., 2017; Foo et al., 2013). According to Gardner, (2008) soil pH is the main factor that influences solubility and availability of plant nutrients in the soil. Our studies also demonstrated that addition of lime and compost alone or in combination greatly decreased aluminum in soil medium (Table 2).
Impact of soil ameliorants on AMF development and plant growth
It’s has been demonstrated that AMF is made up of symbiotic organisms between fungi and plant roots which have numerous benefits to plant growth and development. These fungi can increase the uptake of several essential nutrients such as Phosphours (Smith et al., 2011), Nitrogen and Potassium (Bucking and Kafle, 2015; Sharma et al., 2017), producing plant growth hormones and phosphatase enzyme (Bini et al., 2017; Foo et al., 2013), defending root against soil borne diseases (Bakhtiar et al., 2010 ). AMF also interact positively with coal waste and wood charcoal in the mycorrhizosphere to increased plant growth (Budi and Christina, 2013; Budi and Setyaningsih, 2013). In the present study, the addition of compost (A1B3) slightly decreased AMF roots colonization of C. calothyrsus while when compost combined with lime (A1B4 and A1B5) greatly increased AMF roots colonization of C. calothyrsus (Figure 1). The inhibition of AMF development by compost probably due to compost cannot reduce the Aluminum content in soil media as shown in Table 2, the concentrations of Al-dd are very high (3.37 cmol(+)/kg). These results are in accordance with other studies where it has been shown that high Aluminium content inhibit AMF development (Budi et al., 2020). Several studies showed that there are no positive correlations between roots AMF colonization and plant growth. According to Siqueira and Saggin Júnior (1995), soils with low fertility limit plant development and increase the dependence of plants on mycorrhizal association. Under these conditions, fungi grow more extensively inside the root to support the development and functioning of external hyphae (Sanders et al., 1977). In the present study, the growth (height, diameter and biomass) of C. Calothyrsus were lower when inoculated with AMF compared to plant inoculated with AMF combined with soil ameliorants (Figure 2). The soil medium amended with lime 5.79 g without AMF inoculation (A0B1), increased height, diameter and biomass growth of C.calothyrsus by 147.2%; 110.34% and 30.89%, respectively compared to control plant (A0B0). When lime was combined with AMF inoculation (A1B1), growth responses were greater than without AMF inoculation and increased height, diameter and biomass growth of C.calothyrsus by 556.7%, 270.11% and 165.40% respectively compared to control plant (A0B0). The combination of compost 35 g and lime 5.79 g to the soil growth plant medium followed by AMF inoculation (A1B4) significantly increased height, diameter and biomass growth of C.calothyrsus by 1,106.82%; 563.22% and 222.39%, respectively compared to control plants (A0B0), and is the highest among the others combination treatments (Figure 2). From the present study, it is clearly demonstrated that AMF inoculation synergistically increased C. calothyrsus growth if combined with lime alone or in combination with compost. Phosphorus is an essential nutrient for plant growth but in acid soil medium Phosphorus is bound by Aluminum and cannot be absorbed by the roots of the plant. Lime plays an important role in increasing soil pH and reducing Aluminum toxicity in soil medium (Teshome et al., 2017). Furthermore, iron phosphate can be released through an enzymatic phosphatase process which is released by the AMF (Sharma et al., 2017). Similar results were found if the lime dosage is 10.58 g added to soil medium (Figure 3).The soil medium amended with lime 10.58 g without AMF inoculation (A0B2), increased height, diameter and biomass growth of C. Calothyrsus by 80.12%; 64.37% and 9.14%, respectively compared to control plants (A0B0). If lime were combined with AMF inoculation (A1B2), growth response was greater than without AMF inoculation, and it increased height, diameter and biomass growth of C. Calothyrsus by 582.49%, 312.64% and 166.80%, respectively compared to control plant (A0B0). The combination of compost 35 g and lime 10.58 g to the soil growth medium followed by AMF inoculation (A1B5) significantly increased height, diameter and biomass growth of C. calothyrsusby 1,022.55%, 491.95% and 213.00%, respectively compared to control plants (A0B0), and was the highest among the other combination treatments. As shown in Table 2, the addition of lime 10.58 g to soil medium increased soil pH from 4.27 to 7.25 and Aluminium content was reduced to undetected amount. Soil medium amanded with lime 10.58 g and compost 35 g increased their pH from 4.27 to 7.76 (Table 2), indicating compost also can increase soil pH and reduce aluminum content to undetected amount. From the present study, it is demonstrated that soil medium amended by lime 5.79 g is better than amended by lime 10.58 g for increasing C. calothyrsus growth without AMF inoculation. The addition of lime 5.79 g and compost 35 g to soil medium followed by AMF inoculation increased the growth of C.calothyrsus better than soil amended by 10.58 g and compost 35 g followed by AMF inoculation.
CONCLUSION
Post mining soil with low pH and high Aluminium content could be improved by soil ameliorants lime and compost with appropriate dosage. The soil ameliorant lime increased soil pH to neutral level and increased C. calothyrsus growth. Addition of lime and compost to soil media followed by AMF inoculation increased significantly the growth of C. calothyrsus. These findings imply the prospective and potential use of AMF as biofertilzer and soil ameliorant for the succesful reclamation of post mining land.
ACKNOWLEDGEMENT
The authors thank Directorate for Research and Community Services, Ministry of Research, Technology, and Higher Education, the Republic of Indonesia, for providing the funding of this research. The authors also thank PT Holcim Indonesia Tbk., Sukabumi, West Java, for providing soil materials in this research.
REFERENCES
Anetor,M.O., and E.A. Akinrinde, 2007. Lime effectiveness of some fertilizers in a tropical acid alfisol. Journal of Central Europeean Agriculture, 8(1): 17-24.
Arshi A. 2015. Reclamation of coal mine overburden dump through enviromental friendly method. Saudi Journal of Biological Sciences: 24(2017)371-378 doi: 10.1016/j.sjbs.2015.09.009
Bakhtiar, Y., S. Yahya, W.Sumaryono, M.S. Sinaga, S.W. Budi, andT. Tajudin. 2010. Isolation and identification of mycorrhizosphere bacteria and their antagonistic effects towards Ganoderma boniense in vitro. Journal of Microbiology Indonesia, 4(2): 96-102.
Benidire L, L,Benidire, A. Boularbah. 2020. Impacts of mining activities on soil properties: case studies from Morocco mine sites. SOIL SCIENCE ANNUAL. 71(4), 395–407. https://doi.org/10.37501/soilsa/133011
Bini, D., C. Alcara dos Santos, M.C.P da Silva, J.A. Bonfirm, andE.J.B.N. Cardoso. 2017. Intercropping Acacia mangium stimulates AMF colonization and soil phosphatase activity in Eucalyptus grandis. Scientia Agricola, 2(2): 102-110.
Bucking, H. andA. Kafle. 2015. Role of arbuscular fungi in the nitrogen uptake of plants: current knowledge and research gaps. Agronomy, 5: 587-612.
Budi S.W, and F.Christina. 2013 Coal waste powder amendment and arbuscular mycorrhizal fungi enhance the growth of jabon (AnthocephaluscadambaMiq) seedling in ultisol soil medium.Journal Tropica Soils18, 59-66
Budi S.W. and L.Setyaningsih. 2013. Arbuscular mycorrhizal fungi and biochar improved early growth of neem (Melia azedarach Linn.) seedling under greenhouse conditions JurnalManajenenHutanTropika19, 103-110
Budi, S.W. C.Wibowo,A. Sukendro and H. S. Bekti. 2020. Growth improvement of Falcataria moluccana inoculated with MycoSilvi grown in post-mining silica sand soil amended with soil ameliorants. Biodiversitas, 21(1): 422-427
Clapp J.P, A.H Fitter,and J.W.Merryweather. 1996. Arbuskular mycorrhizas. In: Hall GS, Lasserre P, Hawksworth DL, Editor. Methods for the Examination of Organismal Diversity in Soils and Sediments. Wallingford, Oxon (UK): CAB International.
Chung, Ren-shih, Wu, and Sun-ho. 2008. Effect of corncob compost on plant growth in an acid red soil. Journal Communications in Soil science and Plant Analysis,28(9): 673-683.
Delhaize, E. andP.R. Ryan. 1995. Aluminum toxicity and tolerance in plants. Plant. Phytol. 107:315-321.
Dong, D. M.H Ramsey, and I. Thornton. 1995. Effect of soil pH on Al availability in soils and its uptake by the soybean plant (Glycine max). Journal of Geochemical exploration. 55(1): 223-230
Estefan G, R. Sommer and J. Ryan. 2013. Methods of soil, plant, and water analysis: A manual for the west Asia and north, 3rd ed. ICARDA, Beirut, Lebanon.
Foo, E., J.J. Ross, W.T. Jones, andJ.B. Reid. 2013. Plant hormones in arbuscular mycorrhizal symbioses: an emergingrole for gibberellins. Annals of Botany,111: 769–779.
Gardner F.P.R.B Pearce, and R.L. Mitchell. 2008. Physiology of Crop Plants. Ames (UK): The Iowa University Press.
Ghose, M.K. and S.R.Majee. 2000. Assessment of dust generation due to opencast coal mining – an Indian case study. Environ. Monit. Assess. 61, 255–263.
Husna H.S.W Budi, I.Mansur and C. Kusmana. 2015. Growth response of kayu kuku (Pericopsismooniana (Thw.) Thw) seedling to indigenous arbuscular mycorrhizal fungi inoculation JurnalPemuliaanHutanTanaman9, 131-148
Husna H., I.Mansur,S.W.Budi, F.D.Tuheteru, A.Arif, E.J.Tuheteru, Albasari. 2019. Effects of arbuscular mycorrhizalfungi and organic material on growth and nutrient uptake by Pericopsismooniana in coal mine Asian.Journal of Plant Sciences18, 101-109
Jayani F.M, S.W Budi, andP. Pamoengkas 2018. Response of forest tree species inoculated with AMF and soil ameliorant addition grown in silica sand. Asian J Agri & Biol. 6(4):556-565
Mrabet S.E, L.Ouahmane,A.E.Mousadik, F.Msanda, Y. Abbas. 2014. The effectiveness of arbuscular mycorrhizal inoculation and bio-compost addition for enhancing reforestation with Argania spinosa in Morocco Open Journal of Forestry4, 14-23
Muindi, E.M, J. Mrema, E. Semu, P.Mtakwa, and C. Gachene. 2015. Effects of lime-aluminium-phosphate interactions on maize growth and yields in acid soils of the Kenya highlands. American Journal of Agriculture and Forestry,3(6): 244-252.
Novak, J.M.,J.A. Ippolito, T.F. Ducey, D.W. Watts, K.A. Spokas, K.M. Trippe, G.C. Sigua, andM.G. Johnson. 2018. Remediation of an acidic mine spoil: Miscanthus biochar and lime amendment affects metal availability, plant growth, and soil enzyme activity. Chemosphere,205: 709-718.
O’Connor P.J, S.E.Smith, F.A. Smith. 2001. Arbuscular mycorrhizal associations in the southern Southern Simpson desert. Aust J Bot. 49:493-499 doi:10.1071/BT00014
Ohsowski B.M., K.Dunfield, J.N. Klironomos,M.M. Hart. 2017. Plant response to biochar, compost, and mycorrhizal fungal amendments in post-mine sandpits Restoration Ecology 26, 63-72
Sanders, F.E.,P.B. Tinker, R.L.B. Black, and S.M. Palmerley. 1977. The development of endomycorrhizal root systems. I. Spread of infection and growth-promoting effects with four species of vesicular-arbuscular endophyte. New Phytologist 78: 553-559
Sharma, N., K. Yadav, and A.Aggarwal. 2017. Role of potassium and arbuscular mycorrhizal fungi in alleviation of water stress on Vigna mungo. Environmental and Experimental Biology, 15: 15-24.
Siqueira, J.O. andO.J. Saggin-Júnior. 1995. The importance of mycorrhizal association in natural low-fertility soils. Pp.240-280. In: Anais do Simpósio Internacional sobre Estresse Ambiental. Belo Horizonte 1992: EMBRAPA/ CNPMS, Sete Lagoas.
Smith, S.E., I. Jakobsen, M.Gronlund, andF.A. Smith. 2011. Roles of Arbuscular Mycorrhizas in Plant PhosphorusNutrition: Interactions between Pathways of PhosphorusUptake in Arbuscular Mycorrhizal Roots Have ImportantImplications for Understanding and Manipulating PlantPhosphorus Acquisition. Plant Physiology, 156: 1050-1057.
Teshome, B., T. Tana, N. Dechassa, and T.N. Singh. 2017. Effect of compost, lime and P on selected properties of acidic soils of assosa. Biology Agriculture and Healthcare. 7(5): 34-44.
Wahab, S.A.A. and F.A. Marikar. 2012. The environmental impact of gold mines: pollution by heavy metals. Central European Journal of Engineering,2: 304-313.
Zatta,P., E. Lain, and C. Cagnolini. 2000. Effect of aluminium on activity of Krebs cycle enzymes and glutamate dehydrogenase in rat brain homogenate. Eur.J.Biochem, 267:3049-3055.