DOI: https://doi.org/10.56669/TVKD4051
ABSTRACT
Plant growth promoting bacteria (PGPB) were isolated and evaluated for the control of plant pathogens. Three PGPB isolates, PGPBs 15, 18 and 23, were selected for evaluation as biocontrol agents under screenhouse conditions. Based on dry matter yield, formulated solid-based inoculants of all three isolates were effective as biocontrol agent against Fusarium oxysporum f. sp. cubense in banana. In terms of dry matter yield and external symptoms for the disease, PGPBs 15 and 23 were as effective as Benleaf, a commercial fungicide, in controlling Fusarium oxysporum in banana.
Keywords: PGPB, solid-based inoculant, biocontrol agent
INTRODUCTION
Modern agriculture has traditionally been dependent on the use of synthetic pesticides to control plant pathogens. Increasing environmental damage attributed to widespread use of chemicals has increased research on sustainable and environmentally-friendly ways of plant disease control.
Root-associated bacteria referred to as plant growth promoting bacteria (PGPB) have been known to improve plant growth through the production of plant growth regulators (PGRs), solubilization of nutrients, and production of antibiotics and other metabolites to control plant pathogens. However, selected bacteria that promote growth both under growth room and greenhouse conditions do not always perform well in field trials. The variability of field responses to inoculation with PGPB is compounded by abiotic stresses in the field. Selection of PGPB with tolerance to abiotic stresses would increase the successful crop response to PGPB inoculation. PGPBs inhabit the rhizosphere, the volume of soil under the influence of the plant root system. Inoculation with PGPB which include the genera Pseudomonas, Serratia, Azospirillum and Bacillus has been reported to limit the growth of certain soil-borne pathogens (Kloepper, 1991).
Biological control of soil-borne plant pathogens has been found to be related to the ability of PGPB to synthesize plant growth regulators like IAA, which enhances growth of the root system and the whole plant (Montesinos, et al., 2002). Biocontrol PGPBs prevent the proliferation of soil-borne pathogens through the production of siderophores. The siderophores bind most of the Fe+3 available in the rhizosphere thus preventing the proliferation of pathogens due to lack of iron (O’Sullivan and O’Gara, 1992). PGPB can also induce systemic resistance against plant pathogens (Leeman, et al., 1995) through increased physical and mechanical strength of the cell wall and development of biochemical and physiological changes in plants (Benhamou et al., 1996; Zdor and Anderson, 1992). Biocontrol PGPBs have the capacity to rapidly colonize the rhizosphere, compete for nutrients and sites of pathogen interaction with plants, and synthesize antimicrobial compounds such as bacteriocins and novel antibiotics (Rangarajan et al., 2003; Gutterson, 1990).
Effective control of plant pathogens by PGPB observed in the laboratory or greenhouse is not always evident in field tests. Successful biological control depends on the ability of the biological control agent to tolerate abiotic fluctuations in the soil environment. Soil type, soil temperature, soil moisture, soil pH, carbon and nitrogen content, are among the abiotic factors that influence the efficacy of most biocontrol agents (Stewart, et al. 2010).
METHODOLOGY
In vitro screening for biocontrol activities
Twenty-six PGPB isolates collected from a previous project, were screened in vitro for biocontrol activities against 12 fungal and seven bacterial plant pathogens. The fungal pathogens used were the following:
Colletotrichum gloeosporioides (anthracnose of mango)
Colletotrichum musae (anthracnose of banana)
Bipolaris maydis (corn leaf blight)
Phomopsis vexans (Phomopsis blight and fruit rot in eggplant)
Curvularia inaequalis (leaf spot)
Thielaviopsis paradoxa (trunk rot of palm)
Rhizoctonia solani (sheath disease complex in rice)
Botryodiplodia (canker disease and leaf spot)
Fusarium oxysporum (vascular wilt disease)
Aspergillus versicolor
Penicillium chrysogenum (leaf spot disease of cucumber)
Rhizopus oligosporus
The bacterial pathogens used were the following:
Ralstonia solanacearum (bacterial wilt of potato and tomato; Moko disease in banana)
Erwinia carotovora (tuber soft rot in potato)
Serratia marcescens (cucurbit yellow vine disease)
Pectobacterium carotovorum (stem rot in tomato)
Bacillus subtilis (soft rot of potato tubers)
Xanthomonas (leaf blight of onion; bacterial spot of tomato)
Micrococcus luteus
The PGPB inoculum was prepared from 48 hr cultures grown in 10 ml nutrient broth at 30°C. The bacterial suspension was concentrated by centrifugation at 3,000 rpm for 25 minutes and was adjusted with sterile distilled water to 1x108cfu/ml.
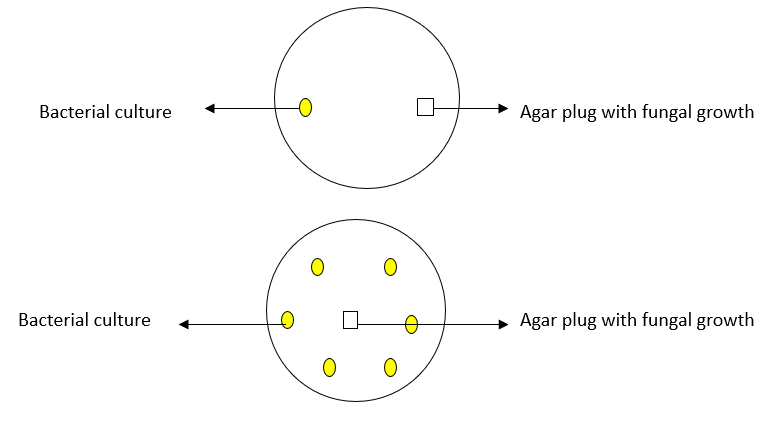
The PDA dual-culture assay set up was used to evaluate the effect of the PGPB on the pathogens. Three drops (0.1 ml/drop~100 µl) of bacterial broth culture and a 6-mm plug from the leading edge of a 5-day old culture of each of the fungal pathogen grown on Potato Dextrose Agar (PDA) were inoculated on opposite sides into 9 cm diameter petri dish containing PDA as shown below. Another set-up was done where 6 bacterial isolates were inoculated with the pathogen at the center of the plate (below). Inhibition was scored by the absence of any contact between the bacterium and the fungi after 5 to 7 days incubation at 28oC.
Evaluation of the effectiveness of selected Plant Growth Promoting Bacteria as biocontrol agents against Fusarium oxysporum in banana (Musa acuminata)
Three selected PGPB isolates, 15, 18 and 23, which exhibited strongest antagonistic activity against some bacterial and fungal pathogens in vitro, were evaluated for biocontrol activity against Fusarium oxysporum, a fungal pathogen which causes Fusarium wilt in banana. The experiment was set up with the following treatments, with four replicates each:
Treatment 1- Inoculated with PGPB 15 solid inoculant and then pathogen
Treatment 2- Inoculated with PGPB 18 solid inoculant and then pathogen
Treatment 3- Inoculated with PGPB 23 solid inoculant and then pathogen
Treatment 4- Inoculated with commercial fungicide (BenleafTM) and pathogen
Treatment 5- Pathogen only
Treatment 6- Control
The PGPB were applied as solid based inoculants. Tissue-cultured banana plantlets were dipped in 15 ml solid based inoculant suspension (100g/1L water) overnight before transplanting into pots. Challenge inoculation was done one month after transplanting. Inoculation with Fusarium oxysporum, was done by applying 5 ml fungal liquid culture at the bases of the plants. Development of disease infection was observed.
Efficacy trials in panama disease infected banana farms
A collaborative trial with the Research and Development Department at Lapanday Agricultural Development Corporation in Davao City, Philippines was established in 2013 to test the efficacy of promising PGPB under field condition with the primary objective of developing a potential biocontrol against Panama wilt disease caused by Fusarium oxysporum f.sp. cubense (Foc). Grand Nain (GN) cultivar of banana was used.
The study was carried out in the nursery, screenhouse and field. The treatments which were laid out in a Completely Randomized Design (CRD) were as follows: PGPB 23 alone, PGPB 23 + Trichoderma harzianum, Trichoderma sp alone, and untreated control.
In the nursery, 100g PGPB inoculant 100mL-1 water and 20g Trichoderma sp. inoculant 100mL-1 water were used. A mixture of the two suspensions were used for the mixed inoculation. Roots of GN meriplants were dipped in the inoculant slurry before planting. The recommended fertilizer was applied. A second inoculation was done one month after planting. After two months the treated plants were tested against Fusarium.
In the screenhouse trial, the pathogen was prepared by using a 7-day old pure culture of Fusarium oxysporum f.sp. cubense. The fungal suspension was standardized at (+/-) 40,000 to 50,000 spores mL-1. The spore suspension was applied at 100 mL plant -1. A 100g 2L-1 suspension of the PGPB inoculant was prepared and 200 to 500 mL was used to drench each plant while Trichoderma sp was applied at 100g plant -1.
An area in the farm with severe PD infection was used for the field trial. Sample plants were planted in the PD eradicated site. Application of the different treatments was done at planting and periodically for five months. Inoculation of the biocontrol agents was done as in the screenhouse trial. Ammonium sulphate was applied at 30g hill-1 at planting. Standard application of commercial fertilizers was also implemented in the trial area. The occurrence of Panama disease was monitored in the sample plants.
RESULTS AND DISCUSSION
In vitro Screening for Biocontrol Activityies
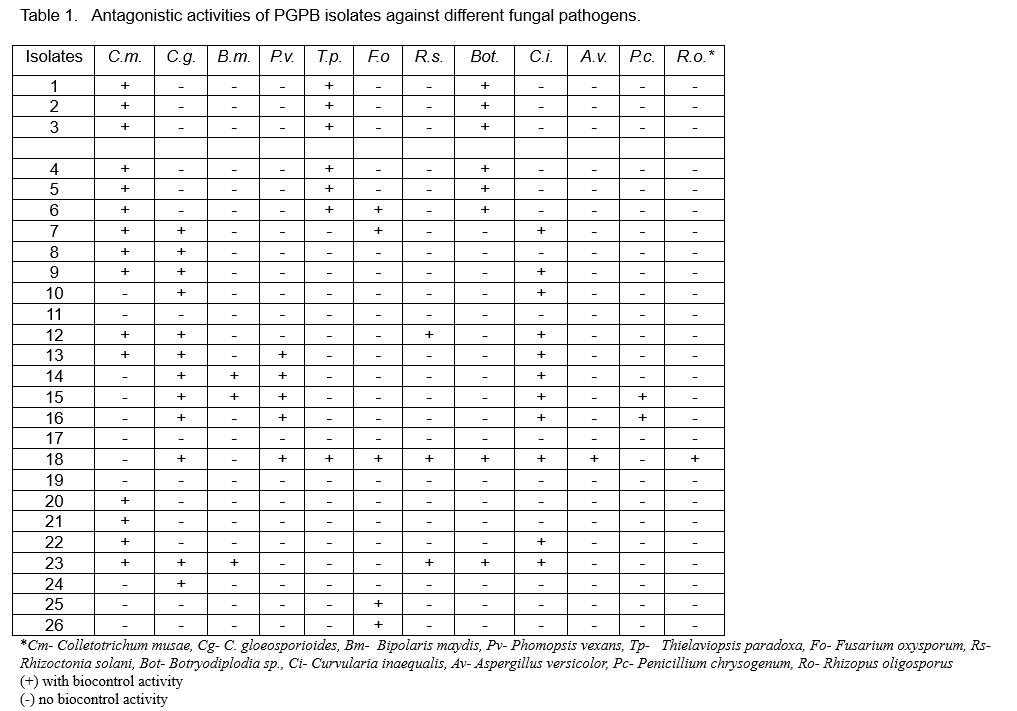
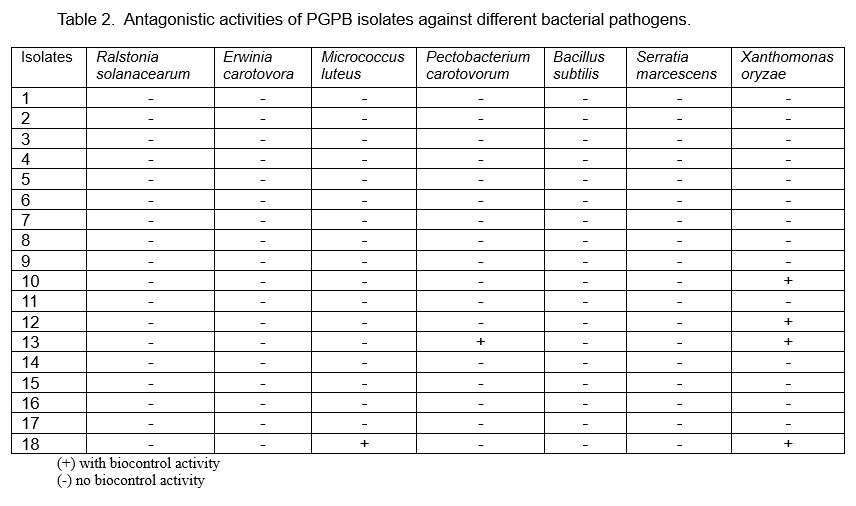
Twenty six PGPB isolates were screened in vitro for biocontrol activity against 12 fungal and seven bacterial plant pathogens. Inhibition was scored by the absence of any contact between the PGPB isolates and the pathogens (Figure 1).
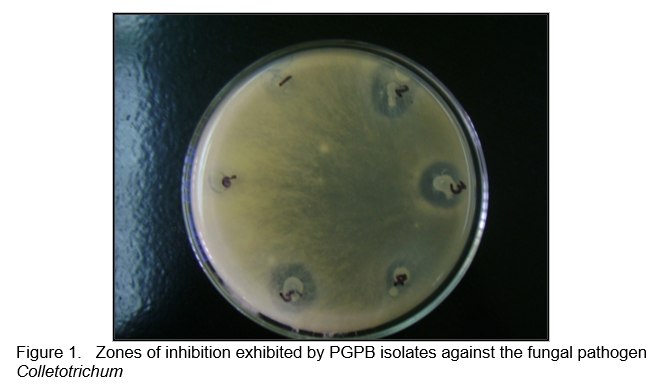
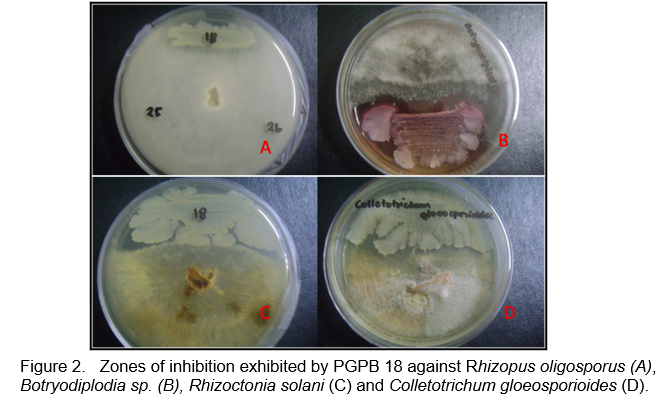
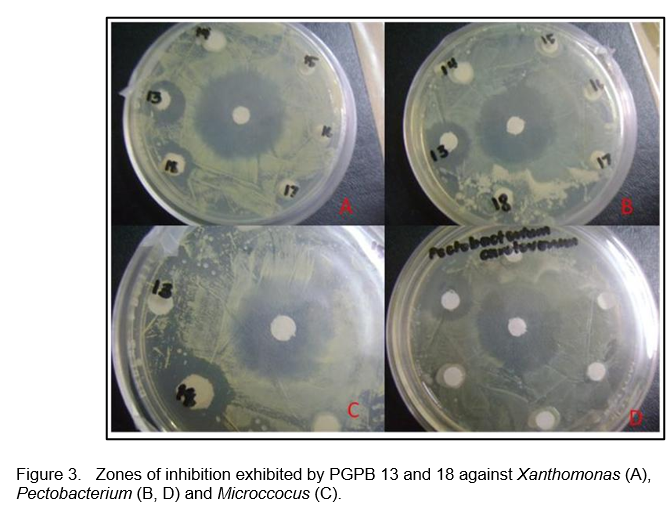
The isolates exhibited biocontrol activity against at least one fungal pathogen (Table 1) and against some bacterial pathogens (Table 2). Most of the isolates showed antagonistic activity against the banana (Colletotrichum musae) and mango (C. gloeosporioides) pathogens and the leaf spot pathogen Curvularia inaequalis. Figures 2 and 3 shows the antagonistic activities of some of the PGPB isolates through the formation of zones of inhibition in the plate assay.
Evaluation of the effectiveness of selected Plant Growth Promoting Bacteria as biocontrol agents against Fusarium oxysporum in banana (Musa acuminata)
PGPB isolates, 15, 18 and 23 were evaluated for biocontrol activity against Fusarium oxysporum, a fungal pathogen which causes Fusarium wilt in banana, in the greenhouse (Figure 4).
In the presence of the pathogen, PGPB inoculated banana seedlings were bigger than the uninoculated plants at 3 weeks after transplanting (Figure 5). Figure 6 shows the banana seedlings at three months after transplanting. The fungus infects banana plants through the roots and invades the vascular tissue (xylem). Test plants treated with the pathogen alone showed external symptoms like gradual wilting, progressive yellowing of the leaves, eventual collapse at the petiole, and longitudinal splitting of the outer leaf sheaths in the pseudostem (Yin et al., 2011). At three months after transplanting the treatment without PGPB had dried up.
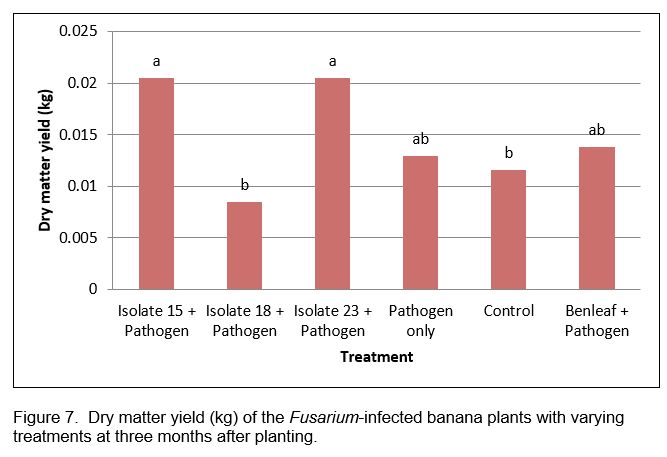
In terms of dry matter yield PGPB 15 and 23 inoculated plants produced the highest dry matter comparable with or even better than the values obtained from Benleaf, the control fungicide (Figure 7). This observation indicates both biocontrol and growth promoting properties of these isolates. The pathogen alone treatment though not significantly different from PGPB inoculation had already dried up at the time of dry matter determination.
Efficacy trials in Panama disease infected banana farms, Davao City
In the nursery the treated plants had significantly higher plant height and leaf area relative to the untreated control. The growth indicators were not significantly different among plants treated with PGPB alone, Trichoderma alone or the combination of the two inoculants. Treated plants inoculated with the pathogen showed 100% PD infection 21 and 23 days after inoculation. However, visual observations showed that PD infection was less severe in plants treated with the combination of PGPB 23 and Trichoderma.
Initial field testing of promising PGPB isolate in the field with severe Panama disease (PD) problem showed that although PGPB 23 reduced PD infection by 70%, it worked best in combination with Trichoderma spp. resulting in only 10% PD infection (Table 3). A 40-day delay in symptom appearance was also observed relative to the untreated control.
The follow up efficacy trial of combined PGPB 23 + Trichoderma spp. inoculation against Panama wilt disease showed that from among 50 test plants per treatment, those inoculated with PGPB 23 + Trichoderma spp. had the lowest % PD infection of 4.55 compared with the untreated control at 30.61% infection at 5 months after planting. Figures 8 and 9 show the crop stand of plants applied with PGPB 23 + Trichoderma spp. and an untreated control.
With this encouraging result obtained in the field, further test is needed by increasing the area to be planted as well as cost analysis on the use of microbial inoculants to combat Panama disease.
Fusarium wilt also called Panama disease is one of the most important and destructive diseases of banana. Bacillus spp. has been identified as potential biocontrol agent for Fusarium wilt (Govindasamy et al., 2011; Tan et al., 2013). The efficacy of Bacillus as biocontrol agent may be attributed to its ability to produce spores which are tolerant to adverse environmental conditions such as heat and desiccation. This project has identified PGPB 23, with 99% similarity to Lysinibacillus fusiformis, a spore-former, as a potential biocontrol agent for Fusarium wilt of banana.





The combination of Trichoderma viride and Bacillus subtilis was reported to be more effective in controlling Fusarium solani in tomato than either one applied alone (Morsy, et al., 2009). The dual treatment reduced the percentage of infection, increased survival rate, and improved fruit yield and nutrient content of tomato. Moreover, dual inoculation enhanced the proliferation the rhizosphere microflora and dehydrogenase activity in the rhizosphere. Dehydrogenase, which is involved in oxidative processes in soils, is a good indicator of microbial activity.
This preliminary study identified promising biocontrol PGPB against Fusarium oxysporum in banana. Further evaluation needs to be done in the field. Moreover, the persistence and rhizosphere competence of the PGPB should be assessed. Studies on the formulation and packaging of the biocontrol PGPB inoculant should also be done.
CONCLUSION
Our results show the potential of plant growth promoting bacteria (PGPB) as biocontrol agents. PGPB can be utilized as low-input biotechnology for sustainable agriculture that is less dependent on external chemical inputs. Biocontrol PGPB has been found effective Fusarium oxysporum in banana. On station and on farm trials indicate the potential of PGPB 23 as a biocontrol agent against Panama disease of banana caused by Fusarium oxysporum f.sp. cubense. Soil type, soil temperature, soil moisture, soil pH, carbon and nitrogen, are among the abiotic factors that affect the efficacy of biocontrol agents (Stewart, et al. 2010). Hence, the promising biocontrol PGPB should be evaluated under varied field conditions. The persistence and rhizosphere competence of the PGPB should be assessed and studies on the packaging of the biocontrol PGPB inoculant should also be done. The promising biocontrol PGPB should be evaluated under varied field conditions. The persistence and rhizosphere competence of the PGPB should be assessed.
ACKNOWLEDGEMENT
We are grateful for the funding support of the Department of Science and Technology- Philippine Council for Industry, Energy, and Emerging Technology Research and Development (DOST-PCIEERD) and the generosity of our farmer collaborators.
REFERENCES
Benhamou, N., R. R. Belanger and T. C. Paultiz. 1996a. Induction of differential host response by Pseudomonas fluorescens in Ri-T-DNA transformed pea roots after challenge with Fusarium oxysporum f.sp. pici and Pythium ultimum. Phytopathology 86:114-178.
Govindasamy V., M. Senthilkumar, V. Magheshwaran, U. Kumar, P. Bose, V. Sharma and K. Annapurna. 2011. Bacillua and Paenibacillus spp.: Potential PGPR for sustainable agriculture. Microbiol. Monographs. 18:333-364.
Kloepper, J. W. 1991. Plant growth-promoring rhizobacteria as biological control agents of soilborne diseases. In: the biological control of plant diseases. Bay-Petersen (ed). FFTC book series no. 42. Food and Fertilizer Technology Center, Taipei, Taiwan.
Leeman, M. .J. A. van Pelt, F. M. Den Ouden, M. Heinesbrock, P.A.H.M. Bakker, and B. Schipperd. 1995. Induction of systemic resistance against furarium wilt of raddish by lipopolysaccharides of P. fluorescens. Phytopathology 85: 1021-1027.
Montesinos, E., A. Bonatera, E. Badosa, J. Frances, J. Alemany, I. Llorente, C. Moragrega. Plant-microbe interactions and the new biotechnological methods of plant disease control. 2002. Int. Microbiol.5:169-175.
Montesinos, E., A. Bonatera, E. Badosa, J. Frances, J. Alemany, I. Llorente, C. Moragrega. Plant-microbe interactions and the new biotechnological methods of plant disease control. 2002. Int. Microbiol.5:169-175.
Morsy, M. E., K. A. Abdel-Kawi and M. N. A. Khalil. 2009. Efficiency of Trichoderma viride and Bacillus subtilis as biocontrol agents against Fusarium solani in tomato plants. Egypt J. Phytopathol. 37(1): 47-57
Rangarajan, S.,.L. M. Saleena, P. Vaudevan and S. Nair. 2003. Biologica suppression of rice disease by Pseudomonas spp. under saline conditions. Plant and Soil 251:73-82
Stewart, A., M. Brownbridge, R. A. Hill and T. A. Jackson. 2010. Utilizing soil microbes for biocontrol. In: Soil Microbiology and Sustainable Crop Production. G. R. Dixon and E. L. Tilston (eds). Springer Science+Business Media B. V. London, New York. pp. 315-371
Tan S., Y. Dong, H, Liao, J. Huang, S. Song, Y. Xu, Q. Shen. 2013. Anatagonistic bacterium Bacillus amyloliquefaciens induces resistance and controls the bacterial wilt of tomato. Pest Manag. Csi. 21. Doi.10.1002/ps.3491.
Yin XM, ZQ Jin, BY Xu, WH Ma, YG Fu, JB Wang. 2011. Characterization of early events in banana root infected with the GFP-tagged Fusarium oxysporum f. sp. cubense. Acta horticulturae 897: 371-376.
Zdor R. F. And A. J. Anderson. 1992. Influence of root coloniaing bacteriaon the defence response in bean. Plant and Soil 140:99-107.
Preliminary Studies on the Biocontrol Property of Plant Growth Promoting Bacteria Against Fusarium oxysporum f. sp. cubense in Banana (Musa acuminata L.)
DOI: https://doi.org/10.56669/TVKD4051
ABSTRACT
Plant growth promoting bacteria (PGPB) were isolated and evaluated for the control of plant pathogens. Three PGPB isolates, PGPBs 15, 18 and 23, were selected for evaluation as biocontrol agents under screenhouse conditions. Based on dry matter yield, formulated solid-based inoculants of all three isolates were effective as biocontrol agent against Fusarium oxysporum f. sp. cubense in banana. In terms of dry matter yield and external symptoms for the disease, PGPBs 15 and 23 were as effective as Benleaf, a commercial fungicide, in controlling Fusarium oxysporum in banana.
Keywords: PGPB, solid-based inoculant, biocontrol agent
INTRODUCTION
Modern agriculture has traditionally been dependent on the use of synthetic pesticides to control plant pathogens. Increasing environmental damage attributed to widespread use of chemicals has increased research on sustainable and environmentally-friendly ways of plant disease control.
Root-associated bacteria referred to as plant growth promoting bacteria (PGPB) have been known to improve plant growth through the production of plant growth regulators (PGRs), solubilization of nutrients, and production of antibiotics and other metabolites to control plant pathogens. However, selected bacteria that promote growth both under growth room and greenhouse conditions do not always perform well in field trials. The variability of field responses to inoculation with PGPB is compounded by abiotic stresses in the field. Selection of PGPB with tolerance to abiotic stresses would increase the successful crop response to PGPB inoculation. PGPBs inhabit the rhizosphere, the volume of soil under the influence of the plant root system. Inoculation with PGPB which include the genera Pseudomonas, Serratia, Azospirillum and Bacillus has been reported to limit the growth of certain soil-borne pathogens (Kloepper, 1991).
Biological control of soil-borne plant pathogens has been found to be related to the ability of PGPB to synthesize plant growth regulators like IAA, which enhances growth of the root system and the whole plant (Montesinos, et al., 2002). Biocontrol PGPBs prevent the proliferation of soil-borne pathogens through the production of siderophores. The siderophores bind most of the Fe+3 available in the rhizosphere thus preventing the proliferation of pathogens due to lack of iron (O’Sullivan and O’Gara, 1992). PGPB can also induce systemic resistance against plant pathogens (Leeman, et al., 1995) through increased physical and mechanical strength of the cell wall and development of biochemical and physiological changes in plants (Benhamou et al., 1996; Zdor and Anderson, 1992). Biocontrol PGPBs have the capacity to rapidly colonize the rhizosphere, compete for nutrients and sites of pathogen interaction with plants, and synthesize antimicrobial compounds such as bacteriocins and novel antibiotics (Rangarajan et al., 2003; Gutterson, 1990).
Effective control of plant pathogens by PGPB observed in the laboratory or greenhouse is not always evident in field tests. Successful biological control depends on the ability of the biological control agent to tolerate abiotic fluctuations in the soil environment. Soil type, soil temperature, soil moisture, soil pH, carbon and nitrogen content, are among the abiotic factors that influence the efficacy of most biocontrol agents (Stewart, et al. 2010).
METHODOLOGY
In vitro screening for biocontrol activities
Twenty-six PGPB isolates collected from a previous project, were screened in vitro for biocontrol activities against 12 fungal and seven bacterial plant pathogens. The fungal pathogens used were the following:
Colletotrichum gloeosporioides (anthracnose of mango)
Colletotrichum musae (anthracnose of banana)
Bipolaris maydis (corn leaf blight)
Phomopsis vexans (Phomopsis blight and fruit rot in eggplant)
Curvularia inaequalis (leaf spot)
Thielaviopsis paradoxa (trunk rot of palm)
Rhizoctonia solani (sheath disease complex in rice)
Botryodiplodia (canker disease and leaf spot)
Fusarium oxysporum (vascular wilt disease)
Aspergillus versicolor
Penicillium chrysogenum (leaf spot disease of cucumber)
Rhizopus oligosporus
The bacterial pathogens used were the following:
Ralstonia solanacearum (bacterial wilt of potato and tomato; Moko disease in banana)
Erwinia carotovora (tuber soft rot in potato)
Serratia marcescens (cucurbit yellow vine disease)
Pectobacterium carotovorum (stem rot in tomato)
Bacillus subtilis (soft rot of potato tubers)
Xanthomonas (leaf blight of onion; bacterial spot of tomato)
Micrococcus luteus
The PGPB inoculum was prepared from 48 hr cultures grown in 10 ml nutrient broth at 30°C. The bacterial suspension was concentrated by centrifugation at 3,000 rpm for 25 minutes and was adjusted with sterile distilled water to 1x108cfu/ml.
The PDA dual-culture assay set up was used to evaluate the effect of the PGPB on the pathogens. Three drops (0.1 ml/drop~100 µl) of bacterial broth culture and a 6-mm plug from the leading edge of a 5-day old culture of each of the fungal pathogen grown on Potato Dextrose Agar (PDA) were inoculated on opposite sides into 9 cm diameter petri dish containing PDA as shown below. Another set-up was done where 6 bacterial isolates were inoculated with the pathogen at the center of the plate (below). Inhibition was scored by the absence of any contact between the bacterium and the fungi after 5 to 7 days incubation at 28oC.
Evaluation of the effectiveness of selected Plant Growth Promoting Bacteria as biocontrol agents against Fusarium oxysporum in banana (Musa acuminata)
Three selected PGPB isolates, 15, 18 and 23, which exhibited strongest antagonistic activity against some bacterial and fungal pathogens in vitro, were evaluated for biocontrol activity against Fusarium oxysporum, a fungal pathogen which causes Fusarium wilt in banana. The experiment was set up with the following treatments, with four replicates each:
Treatment 1- Inoculated with PGPB 15 solid inoculant and then pathogen
Treatment 2- Inoculated with PGPB 18 solid inoculant and then pathogen
Treatment 3- Inoculated with PGPB 23 solid inoculant and then pathogen
Treatment 4- Inoculated with commercial fungicide (BenleafTM) and pathogen
Treatment 5- Pathogen only
Treatment 6- Control
The PGPB were applied as solid based inoculants. Tissue-cultured banana plantlets were dipped in 15 ml solid based inoculant suspension (100g/1L water) overnight before transplanting into pots. Challenge inoculation was done one month after transplanting. Inoculation with Fusarium oxysporum, was done by applying 5 ml fungal liquid culture at the bases of the plants. Development of disease infection was observed.
Efficacy trials in panama disease infected banana farms
A collaborative trial with the Research and Development Department at Lapanday Agricultural Development Corporation in Davao City, Philippines was established in 2013 to test the efficacy of promising PGPB under field condition with the primary objective of developing a potential biocontrol against Panama wilt disease caused by Fusarium oxysporum f.sp. cubense (Foc). Grand Nain (GN) cultivar of banana was used.
The study was carried out in the nursery, screenhouse and field. The treatments which were laid out in a Completely Randomized Design (CRD) were as follows: PGPB 23 alone, PGPB 23 + Trichoderma harzianum, Trichoderma sp alone, and untreated control.
In the nursery, 100g PGPB inoculant 100mL-1 water and 20g Trichoderma sp. inoculant 100mL-1 water were used. A mixture of the two suspensions were used for the mixed inoculation. Roots of GN meriplants were dipped in the inoculant slurry before planting. The recommended fertilizer was applied. A second inoculation was done one month after planting. After two months the treated plants were tested against Fusarium.
In the screenhouse trial, the pathogen was prepared by using a 7-day old pure culture of Fusarium oxysporum f.sp. cubense. The fungal suspension was standardized at (+/-) 40,000 to 50,000 spores mL-1. The spore suspension was applied at 100 mL plant -1. A 100g 2L-1 suspension of the PGPB inoculant was prepared and 200 to 500 mL was used to drench each plant while Trichoderma sp was applied at 100g plant -1.
An area in the farm with severe PD infection was used for the field trial. Sample plants were planted in the PD eradicated site. Application of the different treatments was done at planting and periodically for five months. Inoculation of the biocontrol agents was done as in the screenhouse trial. Ammonium sulphate was applied at 30g hill-1 at planting. Standard application of commercial fertilizers was also implemented in the trial area. The occurrence of Panama disease was monitored in the sample plants.
RESULTS AND DISCUSSION
In vitro Screening for Biocontrol Activityies
Twenty six PGPB isolates were screened in vitro for biocontrol activity against 12 fungal and seven bacterial plant pathogens. Inhibition was scored by the absence of any contact between the PGPB isolates and the pathogens (Figure 1).
The isolates exhibited biocontrol activity against at least one fungal pathogen (Table 1) and against some bacterial pathogens (Table 2). Most of the isolates showed antagonistic activity against the banana (Colletotrichum musae) and mango (C. gloeosporioides) pathogens and the leaf spot pathogen Curvularia inaequalis. Figures 2 and 3 shows the antagonistic activities of some of the PGPB isolates through the formation of zones of inhibition in the plate assay.
Evaluation of the effectiveness of selected Plant Growth Promoting Bacteria as biocontrol agents against Fusarium oxysporum in banana (Musa acuminata)
PGPB isolates, 15, 18 and 23 were evaluated for biocontrol activity against Fusarium oxysporum, a fungal pathogen which causes Fusarium wilt in banana, in the greenhouse (Figure 4).
In the presence of the pathogen, PGPB inoculated banana seedlings were bigger than the uninoculated plants at 3 weeks after transplanting (Figure 5). Figure 6 shows the banana seedlings at three months after transplanting. The fungus infects banana plants through the roots and invades the vascular tissue (xylem). Test plants treated with the pathogen alone showed external symptoms like gradual wilting, progressive yellowing of the leaves, eventual collapse at the petiole, and longitudinal splitting of the outer leaf sheaths in the pseudostem (Yin et al., 2011). At three months after transplanting the treatment without PGPB had dried up.
In terms of dry matter yield PGPB 15 and 23 inoculated plants produced the highest dry matter comparable with or even better than the values obtained from Benleaf, the control fungicide (Figure 7). This observation indicates both biocontrol and growth promoting properties of these isolates. The pathogen alone treatment though not significantly different from PGPB inoculation had already dried up at the time of dry matter determination.
Efficacy trials in Panama disease infected banana farms, Davao City
In the nursery the treated plants had significantly higher plant height and leaf area relative to the untreated control. The growth indicators were not significantly different among plants treated with PGPB alone, Trichoderma alone or the combination of the two inoculants. Treated plants inoculated with the pathogen showed 100% PD infection 21 and 23 days after inoculation. However, visual observations showed that PD infection was less severe in plants treated with the combination of PGPB 23 and Trichoderma.
Initial field testing of promising PGPB isolate in the field with severe Panama disease (PD) problem showed that although PGPB 23 reduced PD infection by 70%, it worked best in combination with Trichoderma spp. resulting in only 10% PD infection (Table 3). A 40-day delay in symptom appearance was also observed relative to the untreated control.
The follow up efficacy trial of combined PGPB 23 + Trichoderma spp. inoculation against Panama wilt disease showed that from among 50 test plants per treatment, those inoculated with PGPB 23 + Trichoderma spp. had the lowest % PD infection of 4.55 compared with the untreated control at 30.61% infection at 5 months after planting. Figures 8 and 9 show the crop stand of plants applied with PGPB 23 + Trichoderma spp. and an untreated control.
With this encouraging result obtained in the field, further test is needed by increasing the area to be planted as well as cost analysis on the use of microbial inoculants to combat Panama disease.
Fusarium wilt also called Panama disease is one of the most important and destructive diseases of banana. Bacillus spp. has been identified as potential biocontrol agent for Fusarium wilt (Govindasamy et al., 2011; Tan et al., 2013). The efficacy of Bacillus as biocontrol agent may be attributed to its ability to produce spores which are tolerant to adverse environmental conditions such as heat and desiccation. This project has identified PGPB 23, with 99% similarity to Lysinibacillus fusiformis, a spore-former, as a potential biocontrol agent for Fusarium wilt of banana.
The combination of Trichoderma viride and Bacillus subtilis was reported to be more effective in controlling Fusarium solani in tomato than either one applied alone (Morsy, et al., 2009). The dual treatment reduced the percentage of infection, increased survival rate, and improved fruit yield and nutrient content of tomato. Moreover, dual inoculation enhanced the proliferation the rhizosphere microflora and dehydrogenase activity in the rhizosphere. Dehydrogenase, which is involved in oxidative processes in soils, is a good indicator of microbial activity.
This preliminary study identified promising biocontrol PGPB against Fusarium oxysporum in banana. Further evaluation needs to be done in the field. Moreover, the persistence and rhizosphere competence of the PGPB should be assessed. Studies on the formulation and packaging of the biocontrol PGPB inoculant should also be done.
CONCLUSION
Our results show the potential of plant growth promoting bacteria (PGPB) as biocontrol agents. PGPB can be utilized as low-input biotechnology for sustainable agriculture that is less dependent on external chemical inputs. Biocontrol PGPB has been found effective Fusarium oxysporum in banana. On station and on farm trials indicate the potential of PGPB 23 as a biocontrol agent against Panama disease of banana caused by Fusarium oxysporum f.sp. cubense. Soil type, soil temperature, soil moisture, soil pH, carbon and nitrogen, are among the abiotic factors that affect the efficacy of biocontrol agents (Stewart, et al. 2010). Hence, the promising biocontrol PGPB should be evaluated under varied field conditions. The persistence and rhizosphere competence of the PGPB should be assessed and studies on the packaging of the biocontrol PGPB inoculant should also be done. The promising biocontrol PGPB should be evaluated under varied field conditions. The persistence and rhizosphere competence of the PGPB should be assessed.
ACKNOWLEDGEMENT
We are grateful for the funding support of the Department of Science and Technology- Philippine Council for Industry, Energy, and Emerging Technology Research and Development (DOST-PCIEERD) and the generosity of our farmer collaborators.
REFERENCES
Benhamou, N., R. R. Belanger and T. C. Paultiz. 1996a. Induction of differential host response by Pseudomonas fluorescens in Ri-T-DNA transformed pea roots after challenge with Fusarium oxysporum f.sp. pici and Pythium ultimum. Phytopathology 86:114-178.
Govindasamy V., M. Senthilkumar, V. Magheshwaran, U. Kumar, P. Bose, V. Sharma and K. Annapurna. 2011. Bacillua and Paenibacillus spp.: Potential PGPR for sustainable agriculture. Microbiol. Monographs. 18:333-364.
Kloepper, J. W. 1991. Plant growth-promoring rhizobacteria as biological control agents of soilborne diseases. In: the biological control of plant diseases. Bay-Petersen (ed). FFTC book series no. 42. Food and Fertilizer Technology Center, Taipei, Taiwan.
Leeman, M. .J. A. van Pelt, F. M. Den Ouden, M. Heinesbrock, P.A.H.M. Bakker, and B. Schipperd. 1995. Induction of systemic resistance against furarium wilt of raddish by lipopolysaccharides of P. fluorescens. Phytopathology 85: 1021-1027.
Montesinos, E., A. Bonatera, E. Badosa, J. Frances, J. Alemany, I. Llorente, C. Moragrega. Plant-microbe interactions and the new biotechnological methods of plant disease control. 2002. Int. Microbiol.5:169-175.
Montesinos, E., A. Bonatera, E. Badosa, J. Frances, J. Alemany, I. Llorente, C. Moragrega. Plant-microbe interactions and the new biotechnological methods of plant disease control. 2002. Int. Microbiol.5:169-175.
Morsy, M. E., K. A. Abdel-Kawi and M. N. A. Khalil. 2009. Efficiency of Trichoderma viride and Bacillus subtilis as biocontrol agents against Fusarium solani in tomato plants. Egypt J. Phytopathol. 37(1): 47-57
Rangarajan, S.,.L. M. Saleena, P. Vaudevan and S. Nair. 2003. Biologica suppression of rice disease by Pseudomonas spp. under saline conditions. Plant and Soil 251:73-82
Stewart, A., M. Brownbridge, R. A. Hill and T. A. Jackson. 2010. Utilizing soil microbes for biocontrol. In: Soil Microbiology and Sustainable Crop Production. G. R. Dixon and E. L. Tilston (eds). Springer Science+Business Media B. V. London, New York. pp. 315-371
Tan S., Y. Dong, H, Liao, J. Huang, S. Song, Y. Xu, Q. Shen. 2013. Anatagonistic bacterium Bacillus amyloliquefaciens induces resistance and controls the bacterial wilt of tomato. Pest Manag. Csi. 21. Doi.10.1002/ps.3491.
Yin XM, ZQ Jin, BY Xu, WH Ma, YG Fu, JB Wang. 2011. Characterization of early events in banana root infected with the GFP-tagged Fusarium oxysporum f. sp. cubense. Acta horticulturae 897: 371-376.
Zdor R. F. And A. J. Anderson. 1992. Influence of root coloniaing bacteriaon the defence response in bean. Plant and Soil 140:99-107.